Scientific calendar August 2022
A rare case of blastic plasmacytoid dendritic cell neoplasm (BPDCN)
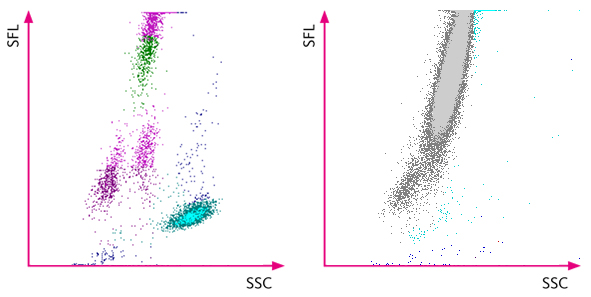
How does the XN-Series indicate a potential presence of malignant cells from the WDF channel?
Atypical Lympho?
Blasts/Abn Lympho?
IG present
Abn Lympho?
Congratulations!
That's the correct answer!
Sorry! That´s not completely correct!
Please try again
Sorry! That's not the correct answer!
Please try again
Notice
Please select at least one answer
Scientific Background
Blastic plasmacytoid dendritic cell neoplasm (BPDCN) is a rare haematologic malignancy derived from the precursors of plasmacytoid dendritic cells which behave like an acute leukaemia. Initially, it was thought to be a kind of lymphocyte-derived cutaneous lymphoma. However, since 2008, the World Health Organisation classifies BPDCN as a distinct disease in the group of myeloid neoplasms [1].
There are no formal studies on the incidence of BPDCN on the general population due to the lack of precise defining criteria prior to the 2008 classification and the constantly changing nomenclature. The disease can occur in all age ranges but usually occurs in elderly, predominantly male patients. The aetiology is unknown so far, and no genetic predispositions or causative environmental factors are identified to date [2]. Findings have been reported suggesting that BPDCN and chronic myelomonocytic leukaemia (CMML) share the same clonal origin [3]. This goes along with the fact that approximately 10–20% of BPDCN patients also have a previous history of haematologic malignancies, e.g. MDS, CML, CMML and AML [2,5].
To a great extent, BPDCN patients show skin involvement in the form of cutaneous lesions, e.g. nodules, plaques or bruise-like filtrates. In fact, these skin lesions are often the reasons why patients seek medical advice [2].
A diagnosis of BPDCN is typically determined based on histopathological and immuno-histochemical examinations. In general, BPDCN can only be diagnosed when the tumour cells demonstrate a blastic morphology, a CD4+ / CD56+ / CD123+ immunophenotype and negativity for lineage specific markers [4,5]. When the bone marrow is involved, thrombocytopenia, neutropenia and anaemia are common findings in the blood analysis as well as hyperleucocytosis and blastosis. Circulating blasts can be morphologically examined in the blood smear and often appear monomorphic, poorly differentiated and intermediate-sized.
The clinical course of BPDCN is highly aggressive with a median overall survival ranging from 12 to 16 months [2]. Considering that the best treatment is not defined yet, the disease can be accounted very serious with bad prognosis. The initial surgical removal of skin lesions followed by conventional radiation therapy seems to show efficacy only for 6–9 months before a systemic relapse occurs. Hence, chemotherapy options are more commonly applied. Nonetheless, conventional intensive chemotherapies, which are normally given for other aggressive leukaemia types, do not appear to be sufficient to cure BPDCN and ensure long-term remissions. For younger patients, known for less aggressive progress of the disease, a chance of significant improvement of survival is given with allogeneic haematopoietic stem cell transplantation (HSCT), especially when performed in first remission. There is some evidence suggesting that specific patients may benefit from autologous HSCT as well, but less empirical data are available to date [2,5].
Targeted therapy options for BPDCN are currently in early clinical trial phases, showing promising efficacy but more evidence is needed. Every treating institution is held to establish and follow a tailored therapeutic approach according to age, sex and comorbidities to improve patient outcomes [2,3].
Patient information
The patient’s initial presentation was with different isolated skin lesions – a rapidly growing nodule on the left arm and three lesions on the back. Histopathological examination showed a morphological and immunohistochemical profile best suited to a ‘blastic plasmacytoid dendritic cell neoplasm’. The treatment regimen for this patient included radiotherapy which led to a reduction of skin lesions and was followed by chemotherapy and antifungal medication for another six weeks. The complete blood count showed fairly normal numerical results afterwards but also demonstrated morphological abnormalities in the WDF scattergram.
Only four months later during a follow-up, the analysis results showed a markedly increased WBC count with blastosis. Palliative treatment was then started because of deterioration of the patient and aggressiveness of the disease.
Result interpretation
During a follow-up in the middle of June, the complete blood count showed normal results for WBC (5.98 x 10³/µL), RBC (4.13 x 106/µL), HGB (13.1 g/dL) and PLT (156 x 10³/µL).
However, an abnormal distribution of the white blood cells in the WDF scattergram was observed, and the XN analyser indicated it by flagging ‘Blasts?’ and ‘Abn Lympho?’. The subsequent manual smear review revealed the presence of blasts.
As a follow-up in early October, the analysis results showed a markedly increased WBC count (70.80 x 10³/µL) with blastosis, anaemia (RBC 1.89 x 106/µL, HGB 5.9 g/dL) and severe thrombocytopenia (PLT&F 14 x 10³/µL).
References
[1] Swerdlow SH et al. (2017): WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues. 4th ed. International Agency for Research on Cancer; Lyon, France.
[2] Pagano L et al. (2016): Blastic plasmacytoid dendritic cell neoplasm: diagnostic criteria and therapeutical approaches. Br J Haematol. 174(2): 188–202.
[3] Brunetti L et al. (2017): Blastic plasmacytoid dendritic cell neoplasm and chronic myelomonocytic leukemia: a shared clonal origin. Leukemia. 31: 1238–1240.
[4] Wang W et al. (2015): Blastic plasmacytoid dendritic cell neoplasm: A case report.
Oncol Lett. 9: 1388–1392.
[5] Laribi K et al. (2020): Blastic plasmacytoid dendritic cell neoplasms: results of an international survey on 398 adult patients. Blood Adv. 4(19): 4838-4848.