Rules to improve reliability
XTRA ARTICLE EDITION 2/2017
Dr Cohen from Geneva University Hospital speaks about how the CBC-O application helps to optimize result reporting for MCHC elevated samples in the haematology lab.
Text: Isabell Spilker
The new ‘CBC-O’ application of the Extended IPU is used to analyse out-of-range MCHC values. Abnormalities are reliably detected and false-positive results are avoided.
Pharmacist and medical biologist Dr Patrick Cohen runs the haematology laboratory at Geneva University Hospital. In this article, he talks about his initial practical experience.

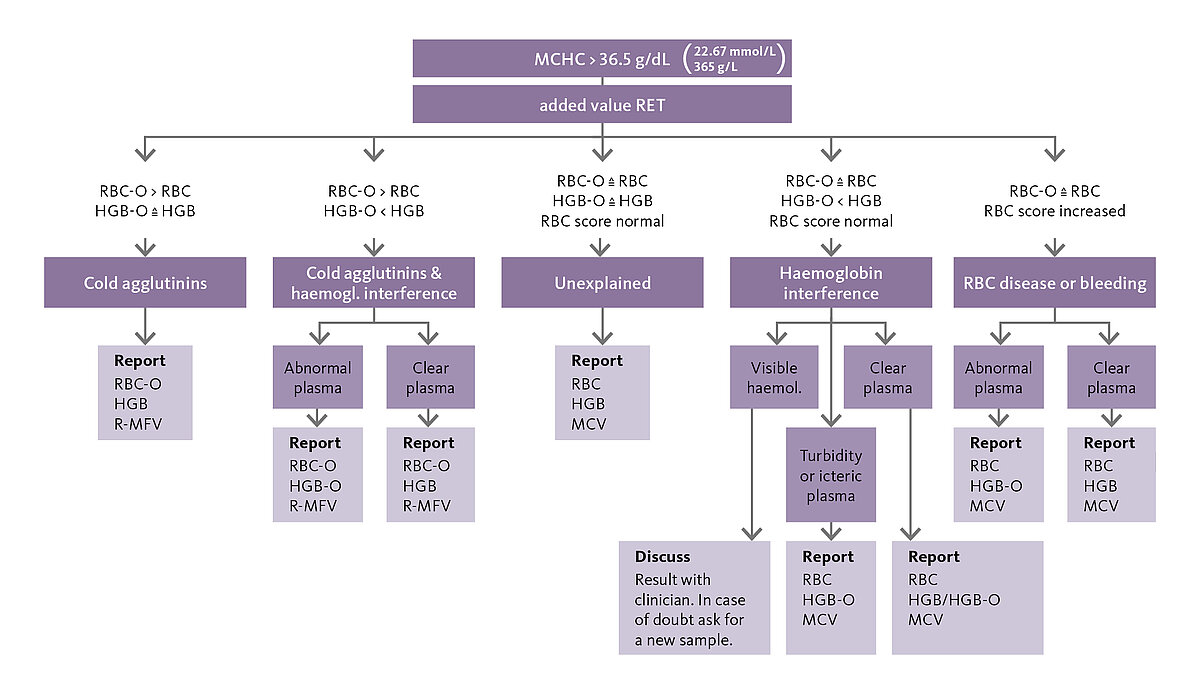
It might not be an impressive acronym, but it has a major impact: The simple acronym ‘CBC-O’ conceals a policy on the management of elevated MCHC (mean corpuscular haemoglobin concentration) values. Increased MCHC values represent rare but time-consuming events in routine laboratory practice that demand proper management. If ignored, spurious results of measured parameters such as the red blood cell (RBC) count, the haemoglobin concentration (HGB), haematocrit (HCT) or the mean corpuscular volume (MCV) may be delivered to the physician with a potentially important impact on the diagnosis and therapy. The pathology-related aetiologies of an elevated MCHC include agglutinins, in vivo haemolysis, and RBC hereditary disease, while interferences such as lipaemia or in vitro haemolysis represent other frequent causes of increased MCHC. By means of Sysmex’s RET channel technology, the CBC-O algorithm can detect the cause of an abnormal result. The haematology laboratory at Geneva University Hospital makes use of the new optional Extended IPU application. Head of the laboratory Dr Patrick Cohen gave us his opinion on the reliability of the diagnosis.
How does the CBC-O application affect your laboratory process?
DR PATRICK COHEN: In order to offer optimised and efficient management of samples displaying an increased MCHC, Berda-Haddad et al. proposed an algorithm in 2016 identifying the different causes of MCHC elevation according to GFHC and ISLH recommendations and a complete process that allows fast and accurate reporting of the results. The CBC-O concept which uses this algorithm allows us to harmonise the process completely, solving the question raised by an increased MCHC in an easy, secured and timely process. It optimises the turnaround time for samples displaying an increased MCHC, and improves standardisation and traceability of the process in our laboratory.
So, does that mean that you now have standardised analysis, above all?
Yes, due to the highly automated function and systematic approach of the CBC-O algorithm, this program improves standardisation and traceability of the process related to high MCHC results management. Indeed, the CBC-O algorithm addresses questions in the same order and proposes a choice between the best results of two channels (impedance/photometry or optical) to solve the problem. This means that all the laboratory technicians will have to do is answer specific predefined questions and approve the proper results suggested by the algorithm, contributing greatly to process standardisation. Furthermore, as any IT-based solution, the CBC-O application allows full traceability of the process.
Apart from more reliable diagnosis: does the CBC-O concept make your daily routine easier?
CBC-O helps us to deliver the correct results faster and reduce the risk of errors – making our results much more reliable. In our laboratory the median time for availability of the result in case of an increased MCHC is around 2 minutes. Besides in terms of implementation, learning to use the CBC-O application in routine is fast and easy for people already working on the Extended IPU unit. The feedback of the laboratory staff concerning this specific item was very positive as they felt more secure and confident in the results they provided to physicians.
Apart from the CBC-O rules, your laboratory uses the biomedical validation criteria based on the GFHC recommendations, which are also defined in the Extended IPU. What advantages do you see here?
They are advantages in many respects: the rules allow all laboratory staff to work in the same way – they all follow the same rules and do the same work. Decisions are not made on a whim at a laboratory level, but are based on official recommendations. The smear rate at our lab used to be extremely high, in part because of home-grown and sometimes contradictory rules. The new rules mean that we have been able to reduce the smear rate in our laboratory. This means much less extra work for staff, so we can avoid fatigue of our staff and focus on difficult cases which require more expertise. This reduces the risk of missing something. The staff also have more time now for further education, for instance.
Do the rules also have a clinical benefit?
I see the clinical benefit as not missing any pathologies, while reducing the number of false-positive smears. If you have time to look left and right and are able to assess all three cell lines in the smear at the same time, the quality of your assessment improves.
Many thanks for the interview!
CBC-O, an application of the Extended IPU, reliably helps to recognise the root cause of an increased MCHC and to achieve correct results with the use of another technology (RET channel).
The RET channel uses fluorescence technology, heats the sample to a certain temperature and measures the intracellular haemoglobin (HBG-O). Compared with the default technologies, many interfering factors affect the results much less in this channel. CBC-O governs the RET analysis, clarifying what has caused the elevated MCHC value and suggesting the use of certain parameters either from the RET analysis (RBC-O, HGB-O) or the impedance/photometric measurement. Its use allows the best possible CBC result to be reported if there is an interference.
The CBC-O concept can only be used on XN analysers with the RET application installed. It accesses the research parameters RBC-O, HGB-O, FRC and R-MFV. The CBC-O algorithm is based on a study by Berda-Haddad Y et al. (2016); onlinelibrary.wiley.com/doi/10.1111/ijlh.12565/pdf